|
These discrete energy steps are what cause atomic line spectra, like the one seen for hydrogen below. The energy is released in the form of light. When the electron moves from a larger higher-energy shell to a smaller lower-energy one it releases energy. When an electron moves to a smaller shell, it releases energy which we observe as light. The amount of energy absorbed or emitted is dependent on the difference in energy between the shells. For an electron to move to another shell it must absorb or release energy. The closer the shell is to the nucleus, the smaller the energy of that shell. Electrons (blue) orbit in circular shells around the nucleus (red).Įach shell has a specific energy level, and electrons cannot exist outside of these orbits. 
In this atomic model, all electrons must exist in a discrete shell and can’t be between shells. The planets are held in orbit by gravitational force and the electrons are held in their orbit by the electrostatic or Coulomb force between the electrons and protons. The electrons orbit around the nucleus similar to how planets orbit around the sun.  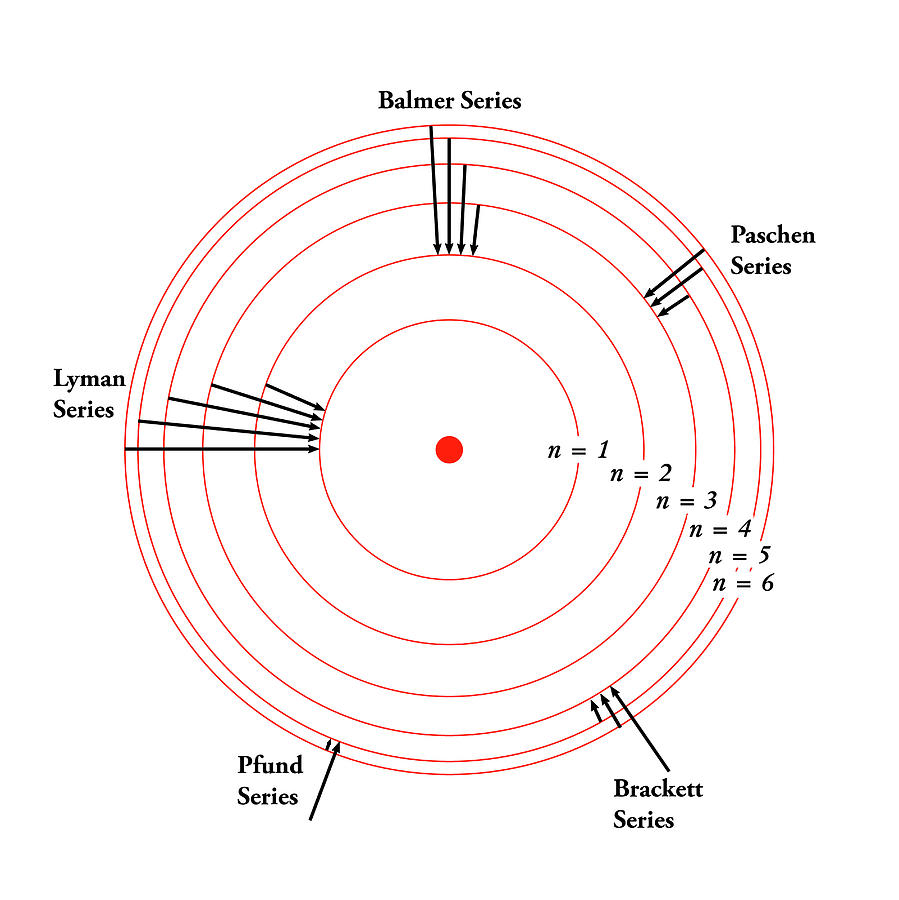
The model is also referred to as the planetary model of an atom. In this model, the electrons travel around the nucleus of an atom in distinct circular orbits, or shells. The model was proposed by physicist Niels Bohr in 1913. In this tutorial, you will learn what the Bohr Model is, how it improved upon previous models of the atom, and what problems the Bohr model fails to solve.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
